Adimmune and NHRI Develop Vaccine for COVID-19 |與國衛院合作 國光生技開發武漢肺炎疫苗
發布時間:
更新時間:
Progress regarding vaccine development has been made, but there is a long way to go. Taiwan-based vaccine maker Adimmune Corporation said it has already finished an analysis of the virus's genetic sequence. The vaccine is expected to be available in 18 months at the earliest.
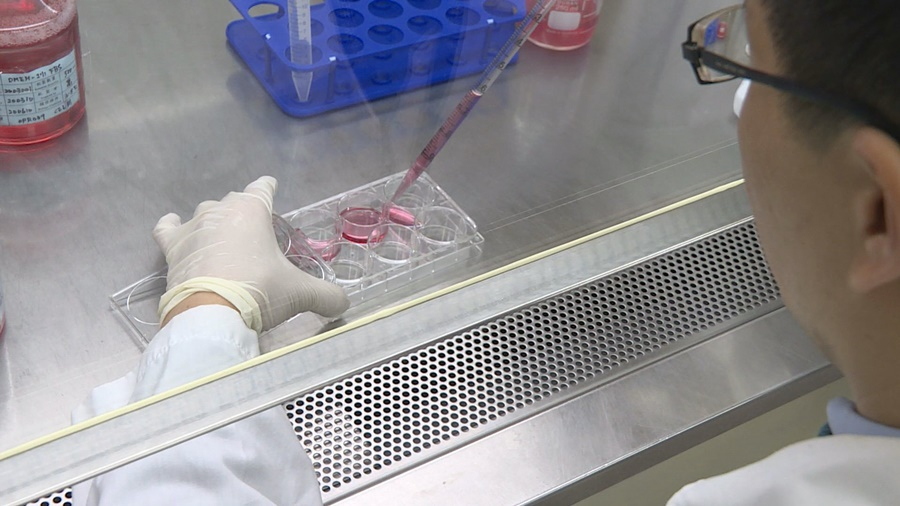
Technicians are carrying out experiements carefully inside the lab. This is the key to vaccine research and development. In response to the timeline of the COVID-19 vaccine, the Adimmune Corporation said it had taken a gene sequence of the coronavirus and applied the protein recombination technique to select the virus protein segment which would allow the human body to produce an immune response. In other words, vaccine development has yielded result.
We take parts of the virus gene and inject the segment to another cell. Then we use the protein recombination to produce proteins and choose the one protein which induces immune response. The one who will stop the virus from growing inside the body.
The Adimmune Corporation pointed out that it will decide on a vaccine candidate in 2 to 3 months which means the company will confirm the decision in May. After the first mass production, the company will then conduct the animal trial between April to June. Clinical trials need to go through the legal process before they can be performed on humans. It is estimated that "1.5 years" is needed before the vaccine could be used on the general public. The Vice President of the Executive Yuan Tsai Chi-chang visited the National Health Research Institutes (NHRI) and offered encouragement to those who are involved in the vaccine development. The NHRI said the most important process in the vaccine development is the human clinical trial stage.
I need to stress that the subjects of the vaccines are not patients but members of the public, therefore safety is the uttermost concern. The vaccine needs to be tested on humans. We must carry out clinical trials on humans and this process could take up to more than a year.
The NHRI has finished the depressant for pneumonia and the synthesis of high-concentration of Remdesivir in grams in just 19 days. The Institute also said that the rapid screening could be considered a treatment indicator. It hopes the virus could be contained by combining the rapid screening, the vaccine and the pharmaceuticals.
技術人員在實驗室,小心翼翼進行實驗,這就是疫苗研發的重要關鍵;武漢肺炎疫情延燒,何時可以有疫苗也備受關注。國光生技表示,肺炎病毒的基因公布後,取出其中一段序列,運用重組蛋白的技術,選取可以讓人體產生免疫反應的病毒蛋白片段,也就是說,疫苗研發已經有了成效。
國光生技總經理 留忠正表示:「(病毒)基因(選取片段),移到其他細胞,然後(透過重組蛋白)直接生產蛋白質,就是一部分(選取)其中一個蛋白質,當然你要找到,那個蛋白質產生的免疫,能抑制病毒在身體的成長。」
國光生技表示,預計在二到三個月內選定候選疫苗,時間大約大五月底可以確定,之後先就一批量產之後,在今年第二季,也就是4-6月進入動物實驗。但何時可以上市,得依照法規進行人體試驗,保守預估,最快還要1年半;而立法院副院長蔡其昌,也前往國衛院慰勉相關人員,國衛院表示,疫苗最重要的程序,就是人體實驗的階段。
國家衛生研究院院長 梁賡義表示:「我要強調,疫苗對象不是病患,而是老百姓,安全非常重要,所以要經過人體試驗,一定要人體試驗,這個進行,可能要一年以上時間。」
國衛院短短19天,就完成肺炎抑制藥物、瑞德西韋公克級高純度合成,也說明快篩也可成為治療指標之一,希望多管齊下,結合快篩,疫苗和製藥方面,能有效控制疫情。