Sanofi Heart Medication Contaminated?|賽諾菲心臟藥"心達舒"冒黑點 緊急回收
發布時間:
更新時間:
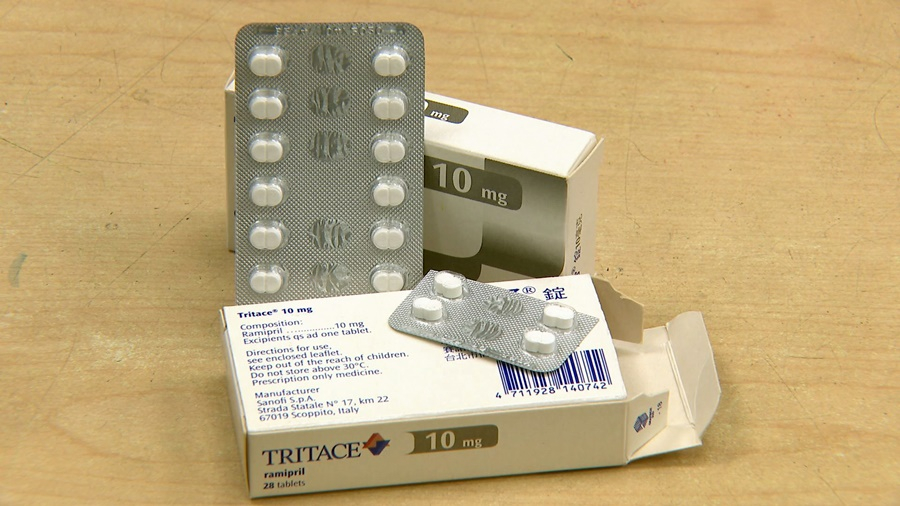
Multinational pharmaceutical company Sanofi was at the center of an impure flu vaccine scandal in 2018. This year, there are issues with its heart medication Tritace. The Food and Drug Administration recently received a report that a portion of 10-milligram Tritace capsules have black spots on them, and believes an entire batch may be tainted. The FDA has asked the pharmaceutical to recall all products from that batch before Jan. 9.
The Food and Drug Administration was recently notified about possibly contaminated 10-milligram Tritace capsules manufactured by multinational pharmaceutical Sanofi. The administration has asked the company to recall all products with the batch number U322 and submit an investigative report.
Perhaps (a worker) accidentally cut his pinky on machinery and the blood got into the capsules.
Tritace, which is prescribed for high blood pressure and heart failure and must be taken regularly, has a 45 percent market share in Taiwan. An estimated 47,000 packages containing 1.3 million capsules from the contaminated batch have already been sold. One doctor says the drug has a long history in Taiwan but there are alternatives patients can take. However, patients should not stop taking the medication without consulting their doctor first.
There are newer alternatives available, so it is actually used less and less in clinical trials. There is no cause for concern. Patients should not stop taking the drug without consulting their doctor. If you stop taking it all of a sudden, your blood pressure could spike.
The administration says it has already asked hospitals and clinics, drug suppliers, and pharmacies to stop using the product and a recall is underway. It is unknown as of now how many of the contaminated capsules have already been ingested. Sanofi says there is little risk of infection from consuming dried blood and concerned patients can return their medication to hospitals or take it to a pharmacy for recycling.
就是這款國際知名藥廠賽諾菲、製造生產的心臟病藥「心達舒」10毫克錠劑,遭食藥署查獲,有部分錠劑外觀出現黑點,疑似有異物混入,因此要求藥廠從即日起,立即回收該批號U322問題藥品,並繳交調查報告。
食藥署簡任技正 黃琴喨表示:「可能是他們(作業員)在操作的當中,不小心被機器割破,一個小手指的一個小傷口,然後血液滲到藥錠裡面。」
食藥署表示,這款藥品主要用於高血壓、心肌梗塞後的心衰竭等,需定期服藥控制的疾病,全台市占率約45%,同批號藥品,約有4萬7千多盒,有130萬顆已售出;而醫師則表示,這款藥品在台灣上市很久,因副作用會口乾舌燥,臨床上有替代藥物,民眾若有疑慮,可回診跟醫師討論,千萬不要自行停藥。
台安醫院心臟內科醫師 林謂文表示:「已經開始有新型的藥物去取代它,所以它其實在臨床上,現在使用量是越來越少,民眾也不用過度擔心,有些使用這個藥物,也不要冒然的停藥,因為有可能會造成血壓會反彈性變高。」
食藥署強調,已請各醫療院所、藥商及藥局,立即停止調劑這款藥品,並進行回收作業,到明年1月9號為止,因此到底有多少顆問題藥品,已被民眾給吃下肚,還要等藥廠送交調查報告才知道。而賽諾菲則發聲明強調,評估因誤食乾燥血液而感染疾病的風險低,若病患對藥品有疑慮,可退回醫院或藥局回收。